Taurine Deficiency Dilated Cardiomyopathy in a Family of Golden Retrievers
Abstract
Dilated cardiomyopathy (DCM) is a disease of the centre musculus that affects both humans and dogs. Certain canine diets have been associated with DCM, but the diet-affliction link is unexplained, and novel methods are needed to elucidate mechanisms. We conducted metabolomic profiling of ix diets associated with canine DCM, containing ≥ 3 pulses, potatoes, or sweet potatoes as primary ingredients, and in the top xvi dog diet brands most frequently associated with canine DCM cases reported to the FDA (3P/FDA diets), and ix non-3P/FDA diets. We identified 88 named biochemical compounds that were college in 3P/FDA diets and 23 named compounds that were lower in 3P/FDA diets. Amino acids, amino acid-derived compounds, and xenobiotics/found compounds were the largest categories of biochemicals that were higher in 3P/FDA diets. Random forest analyses identified the top xxx compounds that distinguished the ii diet groups with 100% predictive accuracy. Iv diet ingredients distinguished the ii diet groups (peas, lentils, chicken/turkey, and rice). Of these ingredients, peas showed the greatest association with higher concentrations of compounds in 3P/FDA diets. Moreover, the current foodomics analyses highlight relationships betwixt diet and DCM in dogs that tin can identify possible etiologies for understanding diet-disease relationships in dogs and humans.
Introduction
Dilated cardiomyopathy (DCM) is a common, progressive, and "largely irreversible" heart affliction affecting humansi. Dilated cardiomyopathy is associated with left ventricular dilation and systolic dysfunction, and usually leads to congestive heart failure or sudden death1,ii. Dilated cardiomyopathy tin consequence from both genetic and ecology causes. Genetic mutations contribute to DCM in humans only known mutations are identified in only 25–40% of those with familial DCM, reflecting substantial gaps in understanding of the causes of DCMiii. In addition, the natural history of DCM in humans is highly variable suggesting that environmental factors may impact disease progression even in DCM with a genetic ground.
This disease does not only occur in humans; DCM is the second virtually common heart affliction affecting pet dogs, with prevalence over fifty% in some breeds, such as the Doberman Pinscheriv,v,half-dozen. DCM in dogs, as in humans, is a serious disease leading to congestive center failure or sudden decease, with survival times typically less than 1 yr subsequently the onset of heart failure and significant echocardiographic improvement unlikely5. Several genetic mutations besides have been associated with DCM in dogs but most dogs with canine DCM do not have a genetic mutation identified7,8,9,10,xi. Even in the Doberman Pinscher, in which two different mutations associated with DCM have been identified, some dogs with DCM have a single mutation, some have both mutations, and some have neither7,8,9. This variable genetic background and a highly variable disease progression, even in dogs with the same mutation, as well support a office for ecology factors in canine DCM.
In addition to genetic causes, DCM can too occur secondary to environmental causes such as direct toxins (e.g., alcohol, chemotherapeutic agents, antibiotics, heavy metals), infectious agents, and nutritional deficiencies12,13,14. Deficiencies of a variety of nutrients, such as thiamine, magnesium, choline, vitamin E and selenium, have been associated with DCM in humans or animal models12,15,16,17. Deficiencies of two amino acid or amino acid-related compounds also can cause a nutritional DCM. Taurine deficiency has been investigated extensively in cats with DCM, where it is known to cause DCM that is reversible with taurine supplementation18,19. In dogs, the role of taurine in DCM is less clear but has been associated with DCM, especially in sure breeds20,21,22,23,24,25,26,27,28,29,thirty,31,32,33,34,35. Deficiency of L-carnitine, an amino acrid-derived molecule, also has been implicated in DCM in dogs and humans36,37. Carnitine belongs to a chemical category of trimethylated fourth amines and imines called "betaines," which have been identified in many foods and linked to human health38,39.
Recent focus on the role of diet in DCM in dogs stems from veterinary reports and Food and Drug Administration (FDA) investigation of a potential link betwixt sure diets and DCM in dogs34,40,41,42,43,44,45,46. Every bit of September, 2020, > 1100 dogs with DCM had been reported to the FDA47. I of the unique characteristics well-nigh the dogs with diet-associated DCM is their outcome. While DCM in dogs is normally a progressive disease with short survival times, multiple studies have shown that dogs with diet-associated DCM take significant echocardiographic comeback and longer survival fourth dimension later on diet modify and medical treatment34,40,44,45,46. The diets reported to exist associated with DCM often are marketed as "grain-free" and oftentimes contain certain ingredients that became part of commercial foods relatively recently (e.one thousand., pulses, potatoes, and sweet potatoes) and lack others (such equally rice or corn). Most of the ingredients that are included in the associated diets are too plant in human being diets, but dogs often eat them in even higher quantities because most dogs eat a single commercial pet nutrient, rather than a variable mixture of multiple foods as humans do. However, intake of certain pet nutrient ingredients such every bit pulses and tubers vary widely amongst human populations, and in some groups represent staple foods that provide substantial sources of protein and energy. Despite ongoing inquiry efforts, agreement of whether diet may be involved in the observed DCM in dogs remains unclear. Standard nutritional analyses of the associated diets take failed to place a causative factor so other, more novel approaches are needed.
One novel arroyo for studying relationships between diet and diseases is metabolomics, which measures modest molecules that tin identify biomarkers of affliction or diet, besides as to assistance delineate mechanistic pathways48,49. While metabolites are typically assessed in plasma or urine, this method tin can as well be used to compare the aforementioned biochemical compounds in foods50, where information technology has been referred to as "foodomics"51. Identifying the biochemical distinguishers of diets associated with DCM in dogs and more traditional dog foods could reveal possible biochemical compounds in nutrient that are contributing to DCM in pet dogs, and too may assistance to identify food ingredients and biochemical compounds that could play a role in human DCM every bit well. The study's primary objective, therefore, was to apply a metabolomics approach to place biochemical compounds that differ between commercial dog foods that have been associated with canine DCM and in more traditional commercial dog foods.
Results
Biochemical compounds differ in two diet groups
A total of 830 biochemical compounds (665 of known identity and 165 of unknown identity) were measured and compared in two diet groups. I grouping (chosen 3P/FDA; n = 9) consisted of diets clinically associated with DCM in dogs, from the top 16 dog nutrient brands fed to dogs with DCM reported to the FDA, and contained ≥ 3 pulses, potatoes, or sweet potatoes in the top 20 ingredients43. The comparing group (non-3P/FDA; n = 9) were diets not associated clinically with DCM in dogs and did not meet the other criteria for the 3P/FDA diets.
Regression coefficients and the log10 P values from the 3P/FDA and non-3P/FDA diet comparison are plotted to show the extent of chemic differences between the diet groups (Fig. 1). Named and unnamed compounds with negative beta coefficients (north = 122, 81.nine%) showed higher concentrations in the 3P/FDA diets, while named and unnamed compounds with positive beta coefficients were lower in 3P/FDA diets (northward = 27, 18.1%; Supplemental Table ane).
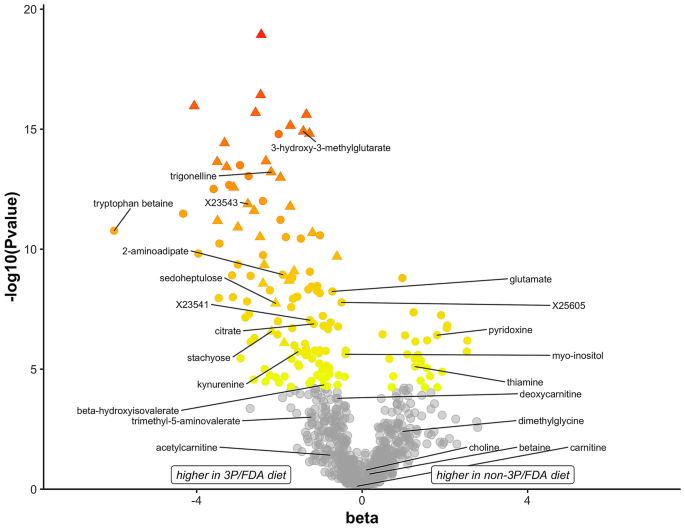
Illustration of 830 biochemical compounds that were significantly dissimilar betwixt diet groups. Diets were assigned to one of two groups: (1) associated with clinical cases of dilated cardiomyopathy (DCM) in dogs; containing ≥ three pulses, potatoes, or sweetness potatoes in the elevation 20 ingredients; and in the top 16 canis familiaris nutrient brands named most oft in DCM cases reported to the FDA (3P/FDA) or (2) non meeting the above criteria (not-3P/FDA). The beta values, where a negative value denotes higher levels in the 3P/FDA diet group, are plotted confronting the negative of logten(P value). Compounds plotted in grayness have P values above the cutoff of five.80E−05, and are considered as non statistically significantly different betwixt diet groups. Triangles indicate compounds that distinguish the two diet groups based on random woods analysis. Those compounds featured in this manuscript are labeled.
Of the compounds that were significantly higher in the 3P/FDA diet group, the largest categories of named compounds were amino acid related (n = 24), and those classed as xenobiotics/plant compounds (n = 20; Table 1). Other categories of compounds that were higher in the 3P/FDA diet group included lipids (n = eighteen), carbohydrates (n = seven), energy metabolism (n = 6), cofactors and vitamins (n = 5), nucleotides (n = five), and peptides (n = 3). A large proportion of compounds that were higher in the 3P/FDA diet group were unnamed (n = 34; Supplemental Table 1). Three of these unnamed compounds were detectable just in the 3P/FDA diet group (X-23541, Ten-23534, and X-25605).
Of the compounds that were significantly lower in the 3P/FDA nutrition group, the largest category of named compounds was cofactors and vitamins (n = 8). Other categories that were lower included xenobiotics/found compounds (n = 6), lipids (n = v), amino acids (north = 3), and carbohydrates (n = 1). There were four unnamed compounds that were lower in 3P/FDA nutrition group.
Using random woods assay, a set of 30 biochemical compounds that distinguished the 3P/FDA and non-3P/FDA diet groups was identified, yielding a predictive accuracy of 100%. Predictive ranks (ane to 30) reflect the biochemical importance of each of these xxx compounds (Supplemental Table 1). Included among these distinguishing compounds are amino acids, cofactors and vitamins, peptides, lipids, nucleotides, carbohydrates (stachyose and sedoheptulose), and 14 unnamed compounds. As plotted in Fig. 1, all 30 compounds distinguishing the ii diet groups were college in the 3P/FDA diet group.
Vitamins were lower in 3P/FDA diets
Seven of the viii vitamins that were significantly lower in 3P/FDA diets were B vitamins: pyridoxine (vitamin B6), thiamine (vitamin B1), folate (vitamin B9), pantothenate (vitamin B5), and riboflavin (vitamin B2) were lower in 3P/FDA diets (Table 2).
Amino acids, amino acid derivatives, and plant-derived compounds differ by nutrition grouping
As indicated above, a number of amino acids and amino acid-related compounds differed significantly betwixt 3P/FDA and not-3P/FDA diets. Tryptophan betaine (525.21 fold), glutamate (1.96 fold), kynurenine (5.66 fold), two-aminoadipate (vi.72 fold; also called 2-aminoadipic acid), pipecolate (12.59 fold), N6,N6-dimethyllysine (four.26 fold), beta-hydroxyisovalerate (2.49 fold; likewise chosen iii-hydroxyisovaleric acrid), and homoarginine (fourteen.02 fold) were among those that were higher in the 3P/FDA diet group (Table 1). Taurine was non significantly different betwixt the diet groups.
Acetyl-D,Fifty carnitine (the D isomer of carnitine; P = 0.038), gamma butyrobetaine (also chosen deoxycarnitine; P = 0.0002), five-aminovaleric acrid betaine (as well called North,N,N-trimethyl-5-aminovalerate; P = 0.001), and dimethyl glycine (P = 0.004) were higher with nominal significance (P < 0.05) in the 3P/FDA nutrition group (Supplemental Tabular array 1). The constitute-derived betaine compounds, trigonelline (N-methylnicotinic acid) and histidine betaine were significantly higher in the 3P/FDA diets (vii.76 fold and 8.30 fold, respectively; Table 1). Carnitine did not differ significantly between the two diet groups.
Other metabolites of interest
Iii additional compounds were also significantly college in the 3P/FDA nutrition group: three-hydroxymethylglutaric acid, myo-inositol, and citrate (Supplemental Table i).
Ingredients are associated with biochemical chemical compound concentrations
Based on biochemical compounds that differed between 3P/FDA and non-3P/FDA diets and differences in some ingredients across diet groups, we reasoned that ingredients could be contributing to the biochemical differences. To investigate ingredient-chemical compound relationships, nosotros first identified "distinguishing ingredients" that differed by five or more domestic dog food products when compared across 3P/FDA and non-3P/FDA diet groups. These 4 ingredients were as follows: (1) peas (nowadays in 9 3P/FDA diets and iv non-3P/FDA diets), (two) lentils (present in half dozen 3P/FDA diets and 1 non-3P/FDA diet), (3) craven/turkey (present in one 3P/FDA diet and viii non-3P/FDA diets), and (4) rice (present in 0 3P/FDA diets and vii non-3P/FDA diets). In the current written report, potatoes and sugariness potatoes were insufficiently represented in either diet type (present in 2 3P/FDA diets and in 1 non-3P/FDA nutrition) to evaluate.
The relationship betwixt each of these iv distinguishing ingredients (peas, lentils, chicken/turkey, and rice) and the biochemical compounds that differed by diet group was visualized by plotting the log2 ratio of the mean concentration of each chemical compound at two ingredient rankings (absent-minded versus loftier) against the P value representing the deviation between the two concentrations at the two ingredient rankings (Fig. 2).
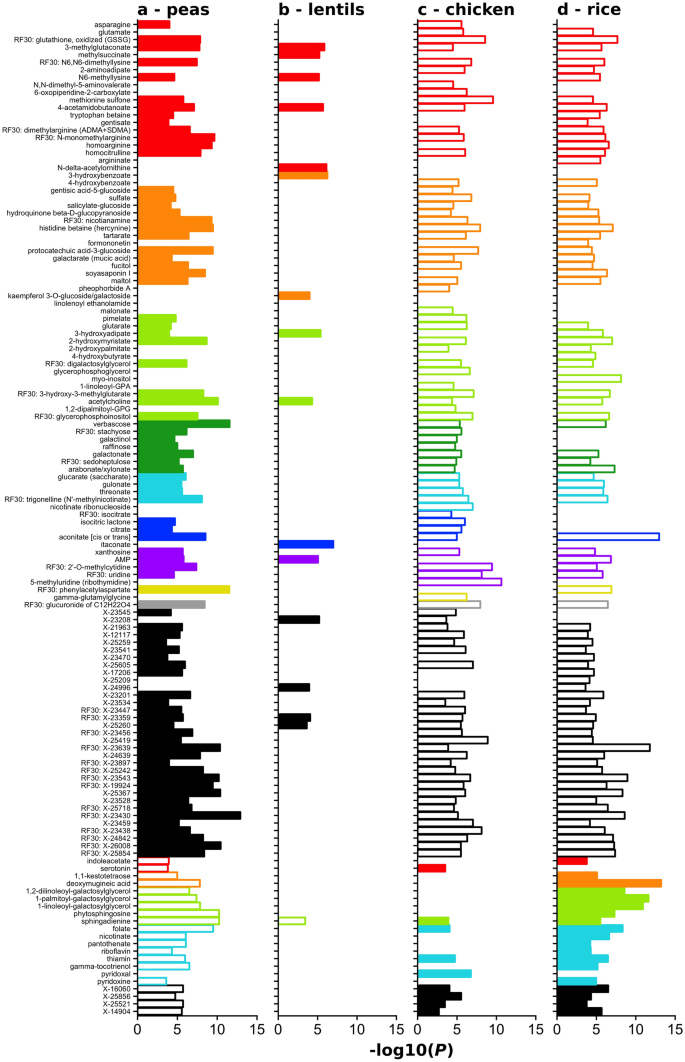
Relationships betwixt four key ingredients and biochemical compounds found to distinguish 3P/FDA from non-3P/FDA diet groups. Panels illustrate the information equally follows: (a) pea; (b) lentil; (c) chicken/turkey; (d) rice. For each of four ingredients that distinguished 3P/FDA and non-3P/FDA diets (i.e., peas, lentils, craven/turkey, and rice), the hateful level of each compound was compared between diets high versus absent in each ingredient (due east.g., diets loftier in peas compared to diets containing no peas). The P value is depicted as − i*(log10[P value]) on the ten-centrality. Solid (filled) confined bespeak a significant positive correlation between compound and ingredient (e.chiliad., diets loftier in peas had significantly higher levels of a chemical compound compared to diets with no peas), while open bars indicate a meaning negative correlation betwixt compound and ingredient. For relationships that do non encounter statistical significance, no bar is plotted. Colors represent the classification of the compounds: cerise, amino acids; orange, xenobiotics and institute-derived compounds; light green, lipid; dark green, carbohydrate; cyan, cofactors and vitamins; blue, energy; violet, nucleotides; xanthous, peptide; grayness, partially characterized; black, unnamed. The designation RF30 indicates a fellow member of thirty compound set determined past random woods analyses to distinguish the 3P/FDA and non-3P/FDA diet groups.
Figure 2 illustrates the compounds that are college and lower in the 3P/FDA diet group compared to the non-3P/FDA diet grouping. Solid (filled) horizontal bars indicate a positive relationship betwixt an ingredient and a compound. When all 4 distinguishing ingredients are plotted (Fig. 2), the ingredient-chemical compound relationship for peas shows more than solid bars for peas compared to the other ingredients, supporting the possibility that peas contribute to college concentrations of these biochemical compounds. In contrast to peas, rice and chicken/turkey are associated primarily with open bars, indicating lower amounts of the compounds in association with these ingredients. Compared to peas, ingredient-compound associations for lentils are fewer, only in a similar direction as peas.
The last 21 compounds of Fig. ii (offset with indolacetate) illustrate the compounds that are lower (open bars) in the 3P/FDA diet group compared to the not-3P/FDA diet grouping and their relationships to the same 4 ingredients. Peas were associated with significantly negative ratios (i.e., lower concentrations of the compound; open bars) for 20 compounds. Lentils are associated with lower concentrations of one compound that is similarly associated with peas. Chicken/turkey and rice are both positively associated with compounds (9 and 19, respectively) that are lower in the 3P/FDA diet group.
These results suggest that peas represent the ingredient contributing the greatest differences betwixt 3P/FDA and not-3P/FDA diets, and that they are associated with higher concentrations of many compounds (88 named biochemical compounds were significantly higher in the 3P/FDA group and 23 named biochemical compounds that were significantly lower in the 3P/FDA grouping). Generally, compounds that were college in the high pea diets are lower in the high chicken/turkey or high rice diets.
Discussion
The current study identified a substantial number of biochemical differences in the 3P/FDA diets, which accept been associated with DCM in pet dogs, compared to non-3P/FDA diets34,40,41,42,43,44,45,46. Most (81.nine%) biochemical compounds that differed between diet groups were higher in 3P/FDA diets compared to non-3P/FDA diets, and four distinguishing ingredients, peas, lentils, craven/turkey, and rice, which also differed across the diet groups, appear to contribute to these differences. Peas and, to a lesser degree, lentils appear to exist a possible primary source for the biochemical compounds plant to exist significantly higher in 3P/FDA diets. While we cannot found with certainty if any of these compounds and ingredients are causal for illness, the findings support peas as a leading possible ingredient associated with nutrition-associated DCM in dogs. In general, diets containing peas were commonly associated with compounds being higher in the 3P/FDA diets, rather than with compounds that were lower or deficient in these diets.
One possible hypothesis regarding how the 3P/FDA diets may contribute to DCM posits that these diets could be insufficient in fundamental nutrients that lead to disease. With respect to food insufficiency, several compounds that are relevant to cardiac metabolism, including B vitamins and related compounds, were lower in 3P/FDA diets. B vitamins are co-factors in numerous reactions relevant to cardiac metabolism. Because vitamins B6 and B12, for example, are co-factors in carnitine and taurine synthesis (both of which are important for normal myocardial role), a deficiency or insufficiency in B vitamins potentially could contribute to DCM52,53. B vitamins are estrus-sensitive, so the results could be related to the corporeality of B vitamins included in the formulations by individual manufacturers and to levels remaining later extrusion. For case, a study of canned true cat foods that were all labeled as consummate and counterbalanced found that 12/90 of the diets were beneath the Association of American Feed Control Officials minimums for thiamine. Pre- and post-manufacture storage conditions also could affect B vitamin levels54. Taurine and carnitine, however, were not significantly dissimilar between the diet groups. Thiamine (vitamin B1) deficiency as well can cause DCM directly12,55. Investigation of potential mechanisms related to B vitamins will require additional analyses, including metabolomics studies in dogs with diet-associated DCM.
In dissimilarity to the small number of compounds that were significantly lower in the 3P/FDA diet grouping, a large set of biochemical compounds were higher. Within the set of compounds that was significantly higher, the categories of amino acids, betaines, and xenobiotics/establish-based compounds are well represented. Although nutrient deficiencies are more plainly suggested when dietary nutrients are lower, such equally the B vitamins noted above, deficiencies as well can be created indirectly by excess or unbalanced nutrient components that interfere with the normal absorption of a nutrient in the alimentary canal. For example, dietary fiber could alter the digestibility and bioavailability of essential nutrients that appear to be adequately supplied in the diet, contributing to a deficiency56. A detail nutrient also could appear to be adequate in the diet just it may not be bioavailable to the animal in a particular form (east.chiliad., organic vs. non-organic sources of zinc, copper oxide vs. copper sulfate)57,58. Anti-nutritional factors naturally nowadays in certain ingredients also could theoretically play a role, although most should be inactivated past heat during the manufacturing process.
In the current study, the over-represented categories of amino-acid related compounds, betaines, and xenobiotics/found-based compounds too could be contributing to deficiencies in molecules that are essential to heart function, such every bit carnitine. Carnitine is disquisitional in cardiac metabolism through its facilitation of the transport of long concatenation fatty acids from the cytosol and across the mitochondrial inner membrane for beta-oxidation. Carnitine itself did not differ beyond the two diet groups, just other amino acid-related compounds that were higher in the 3P/FDA diets could influence carnitine metabolism. Specifically, several compounds that were previously shown to change carnitine transport could touch on carnitine bioavailability to cardiac tissue. The high affinity carnitine transporter, primarily designated as SLC22A5 and formerly called OCTN2 (organic cation transporter novel 2), was identified and characterized over two decades ago59, and its connection to DCM described over the last decade60. Grube et al. noted that OCTN2 expression in heart tissue was lower in man patients with DCM, and that genetic variants of SLC22A5 caused heart failure through reduced uptake of carnitine. Several compounds institute to be college in 3P/FDA diets in the current report have been demonstrated to reduce L-[3H] carnitine transport into the myocardium through inhibition of the transporter59,61. These inhibitors include the D isomer of carnitine, acetyl-D,50 carnitine, gamma butyrobetaine (deoxycarnitine), choline, and betaine59,61, and the first 2 of these carnitine transport inhibitors were college in 3P/FDA diets. A third compound, 5-aminovaleric acrid betaine (N,N,N-trimethyl-5-aminovalerate) was shown to inhibit beta-oxidation of fatty acids in mouse cardiomyocytes, besides through inhibition of the carnitine transportertwoscore and was besides higher in 3P/FDA diets in the current study. These findings back up the possibility that 3P/FDA diets supply biochemical compounds that limit carnitine bioavailability at the level of the mitochondria, interfering with fatty acid oxidation and reducing the heart's energy supply. Altogether, these relationships support i potential means by which 3P/FDA diets contribute to DCM, although others are possible through various mechanisms that affect myocardial metabolism.
In add-on to deficiencies, other possibilities through which the 3P/FDA diets could cause DCM are compounds that have directly toxic event on the myocardium. For instance, plants such as almonds, fruits, and beans can release cyanide, which is cardiotoxic62. In the current written report, a big number of "unnamed" compounds differed between 3P/FDA and non-3P/FDA diets. Additional work is needed to identify them and whatever potential role they may play in heart office. We are also unable to exclude the possibility that added or naturally occurring chemicals (due east.g., pesticides, mycotoxins, and heavy metals) are present as toxic contaminants in the foods, but were not detected through metabolomics.
The current projection focuses on small molecules in canis familiaris foods that have been associated with DCM, only at least two relevant studies accept assessed metabolites in samples of humans with and without DCM63,64. Because many compounds are derived from foods and could impact disease risk without undergoing metabolic conversion, findings from these previous studies are relevant to understanding nutrition-disease relationships. In these published reports, half dozen compounds were significantly higher in human DCM patients63 or in humans with increased illness severity64: glutamic acid, 3-hydroxymethylglutaric acid, myo-inositol, 4-acetamidobutanoate, kynurenine, and gamma-glutamylisoleucine. In the current study, all six of these compounds also were significantly college in the 3P/FDA diets compared to the non-3P/FDA diets. In dissimilarity, college levels of three compounds—3-hydroxyisovaleric acrid (as well chosen beta-hydroxyisovalerate), aminoadipic acid, and citric acid—were associated with protection against DCM in human patients64. These three compounds were all higher in 3P/FDA (DCM-associated) diets in the current study. These findings reinforce the need to follow up this foods-based research in dogs and humans, including feeding studies in dogs with and without DCM, to sympathize the relationships between foods, biochemical compounds, and heart disease in both species.
Although the primary objective of this metabolomics approach was to place biochemical compounds that distinguished 3P/FDA and non-3P/FDA diets, the bear on of ingredients in contributing to these differences was besides of interest. Peas emerged equally one ingredient that differed across the two nutrition groups, and was too positively and strongly associated with many compounds that were higher in 3P/FDA foods. Several independent studies, including dietary interventions that used metabolomics analyses, previously confirmed associations between peas and several compounds (trigonelline, dimethylglycine, and tryptophan betaine) that our study identified every bit college in the 3P/FDA diets65,66,67. Although trigonelline (nicotinic acrid Due north-methylbetaine) can be classified as a product of niacin (vitamin B3) metabolism in mammals, it also occurs in many plants67. Trigonelline was identified as a mark of pea intake in a human feeding study65. A 2d, randomized, crossover report in people identified tryptophan betaine, trigonelline, and dimethylglycine as markers of legume intake66. It is unclear whether whatsoever of these or other compounds found in high levels in peas could accept negative effects on the center when fed in large amounts, but this warrants further investigation. Other compounds that distinguished 3P/FDA and non-3P/FDA diet groups, such as the tetrasaccharide stachyose, are found in legumes but are non specific to peas68,69. Previous reports on ingredients that contribute to DCM in dogs have implicated pulses in general or specific pulses such as lentils or peas34,35,43,44. As of April thirty, 2019, 89% of DCM-associated diets reported to the FDA independent peas and 93% of diets independent peas or lentils43. In the current study, lentils were not equally strongly associated with compounds that were significantly different between diet groups although their patterns were like to those of peas. Our results practise not support a office for other pulses (e.one thousand., chickpeas), which were present much less frequently than peas in the diets we analyzed, just additional inquiry is warranted to fully evaluate the associations with pulses other than peas.
Numerous limitations must be acknowledged. While 18 dog food products were analyzed (nine of each nutrition group), the selection was based on diets that were associated with clinical cases of DCM seen by the authors. All diets in the 3P/FDA group contained ≥ iii pulses, potatoes, or sweet potatoes in the pinnacle xx ingredients and all were in the top xvi domestic dog nutrient brands named virtually oft in DCM cases reported to the FDA43. However, information technology is of import to note that there was some subjectivity in the pick and the diets in both groups represent a small number of products relative to all dog foods available in the U.s. marketplace. Although informative, the private selections and even the nutrition categories may non exist the optimal categorization and additional enquiry volition keep to add together to the knowledge base to refine the pattern of future studies and clinical recommendations. Information technology is possible that the causative or contributing factor(southward) in the 3P/FDA diets was ane of the many unnamed compounds identified or a factor was non measured in the electric current metabolomic analysis. That only two food samples were analyzed from each selected product, and not all samples were obtained from two unlike bags of food. (i.e., in some cases, both samples were from a unmarried handbag) is another limitation. Some of the diet samples were obtained directly from owners of dogs with DCM; most of these diets had been opened and stored for variable periods of time, first by the owner and then by the investigators, then this could innovate variability and could bias the 3P/FDA diets in having lower levels of certain nutrients that could degrade equally a result of variable storage conditions. In add-on, the relative rank for each ingredient (absent, low, moderate, high) was based on the ingredient'southward position on the food label and was not based on quantitative information on the verbal amount of peas or other ingredients that are present in each individual product. Finally, all pea fractions (e.yard., peas, pea protein, pea starch, pea fiber) were classified in the same manner and information technology is non nevertheless known whether pea fractions accept similar effects.
The current study used a metabolomics approach to foods ("foodomics") to place molecular and potential ingredient sources that differentiate 2 diet groups reported to modify DCM take a chance in pet dogs. Biochemical compounds that differed past nutrition group included amino-acid related compounds, carnitine-related molecules, the set of methylated compounds referred to equally betaines, and a variety of unnamed xenobiotics and constitute compounds, and we hypothesize that ane or more of these compounds may contribute to DCM in dogs. The hypotheses generated will require follow-up interventions with metabolomics in dogs to constitute causality and provide definitive nutrition-disease evidence. Finally, while this report focuses on a specific form of heart illness in dogs, it demonstrates the usefulness of investigating relationships between biochemical compounds, food ingredients, and diseases such as DCM.
Methods
Biochemical analyses were limited to commercial dog foods. The project did non include any interaction with or any experiments on vertebrate animals. All analyses were carried out in accordance with relevant guidelines and regulations including validation by Clinical Laboratory Comeback Amendments (CLIA), ISO 9001certification and accreditation by the College of American Pathologists and the New York State Department of Health Clinical Laboratory.
Diets
Nine dry (extruded) dog foods that clinically appeared to be associated with DCM in pet dogs were selected for analysis. These 9 diets all contained at to the lowest degree three occurrences of pulses, potatoes, or sugariness potatoes in the height 20 ingredients and were in the top 16 dog food brands fed to dogs with DCM reported to the FDA in the agency'southward update in June, 2019 (3P/FDA diets)43. In addition, nine dry dog foods that met Globe Minor Animal Veterinary Association recommendations70, were not, in the authors' feel, associated clinically with DCM in dogs, and did non meet the other criteria for the 3P/FDA diets were selected for analysis as controls (Non-3P/FDA diets). Ii samples from each production were obtained and so that each production could exist tested in duplicate. Where possible, samples that were obtained straight from owners of dogs with DCM were used for the analysis. For these diets, just a single bag of nutrient was used and two samples from separate parts of the pocketbook were selected. Most of these diets had been opened by the owner and stored for variable periods of time, first by the owner and then by the investigators. When diets beingness fed to individual dogs were not available, two new bags from the aforementioned lot were purchased and one sample from each bag was collected for analysis. All samples were labeled with a code and so the identity was unknown to the laboratory. Samples were shipped overnight to a commercial laboratory (Metabolon, Inc., Morrisville, NC).
Metabolomic assay
Metabolic profiling of two samples from each of eighteen dog food products was conducted by Metabolon, Inc. using standardized methods. Biochemical compounds were quantified using ultra‐high‐operation liquid chromatography–tandem mass spectroscopy and identified by comparison to a reference library of 4500 purified standards containing retention time, molecular weight, mass–charge ratio, and mass spectroscopy spectral information. A full of 830 biochemical compounds were detected, identified, and met quality control requirements according to standard protocols of the commercial laboratory. All identified compounds, both named and unnamed, were included in the analyses.
Ingredient ranking
Diets' ingredient lists varied both within and between nutrition groups (i.e., 3P/FDA and non-3P/FDA). While some ingredients were nowadays only in one diet group (for instance, grains were present merely in the non-3P/FDA diet group), other ingredients were shared beyond both diet groups. Ingredient positions on domestic dog food labels, similar human food labels, indicate relative amounts of each ingredient by weight. To facilitate analyses of the relationships betwixt ingredients and biochemical compounds, a relative rank for each ingredient (absent, low, moderate, high) was assigned by one of the authors (LMF, a veterinary nutritionist) based on the ingredient's position on the food characterization.
Identification of distinguishing ingredients
Dog foods included a diverseness of ingredients, with up to 57 ingredients in a single diet. When all the different ingredients beyond the 18 unlike diets were added, several hundred ingredients were represented, some of which are contained but in a single food or a few foods, while others were common within a diet group. Nosotros identified major distinguishing ingredients that differed by five or more domestic dog foods across the 2 diet groups. For instance, rice was present in 7/ix non-3P/FDA diets and 0/9 3P/FDA diets, yielding a difference (7–0) of 7. Mineral supplements and probiotics were excluded from ingredients analysis. Ingredients that differed by v or more canis familiaris foods when compared across 3P/FDA and non-3P/FDA diet groups were considered "distinguishing ingredients."
Statistical analyses
Linear regression was used to assess associations between the 3P/FDA and non-3P/FDA diet groups and the individual biochemical compounds present in the foods. A conservative Bonferroni correction for 830 biochemical compounds was applied to determine statistical significance (0.05/830 = 0.00006). P values < 0.05 and ≥ 0.00006 were considered to exist nominally significant.
Random woods nomenclature was applied to biochemical compounds in 3P/FDA and non-3P/FDA diets to identify the top 30 compounds that yielded a predictive accurateness of 100%. These compounds were identified through "importance" rank ordering that was generated through random permutation.
Analysis of the relationship between ingredients and biochemical compounds that differed significantly between 3P/FDA and non-3P/FDA diets was assessed past pair-wise comparison of compounds when each of iv distinguishing ingredients was 'absent' compared to when each ingredient was ranked 'high'. Statistical significance for ingredient-compounds analyses was conservatively corrected past multiplying the number of compounds tested by the number of ingredients tested (four). Specifically, for named compounds that were higher in 3P/FDA foods (due north = 88 compounds) the threshold P threshold was calculated every bit 0.05/(88 × iv) to yield P = 0.0001. For named compounds that were lower in 3P/FDA foods (n = 23), the P threshold was calculated as 0.05/(23 × 4) to yield P = 0.0005. Similar methods were applied for unnamed compounds. For unnamed compounds that were higher in 3P/FDA foods (n = 34), the threshold P value was 0.0004 and for unnamed compounds that were lower in 3P/FDA foods (n = 4), the threshold P value was 0.003. Statistical analyses for biochemical compounds and ingredients were conducted in SAS 9.4.
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to ongoing research simply are available from the corresponding author on reasonable request.
References
-
Maron, B. J. et al., for the American Heart Clan, Council on Clinical Cardiology, Heart Failure and Transplantation Commission, Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups, and Quango on Epidemiology and Prevention groups. Gimmicky definitions and nomenclature of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Enquiry and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Quango on Epidemiology and Prevention. Circulation. 113, 1807–1816 (2006).
-
Japp, A. G., Gulati, A., Cook, S. A., Cowie, K. R. & Prasad, Southward. One thousand. The diagnosis and evaluation of dilated cardiomyopathy. J. Am. Coll. Cardiol. 67, 2996–3010 (2016).
-
Rosenbaum, A. N., Agre, K. East. & Pereira, Northward. L. Genetics of dilated cardiomyopathy: Practical implications for centre failure management. Nat. Rev. Cardiol. 17, 286–297 (2020).
-
Calvert, C. A. et al. Association between results of ambulatory electrocardiography and evolution of cardiomyopathy during long-term follow-upwards of Doberman Pinschers. J. Am. Vet. Med. Assoc. 216, 34–39 (2000).
-
Martin, Thousand. Due west. South., Stafford Johnson, M. J. & Celona, B. Canine dilated cardiomyopathy: A retrospective study of signalment, presentation and clinical findings in 369 cases. J. Small-scale Anim. Pract. fifty, 23–29 (2009).
-
Wess, G. et al. Prevalence of dilated cardiomyopathy in Doberman Pinschers in various historic period groups. J. Vet. Intern. Med. 24, 533–538 (2010).
-
Meurs, K. M. et al. A splice site mutation in a gene encoding for PDK4, a mitochondrial protein, is associated with the development of dilated cardiomyopathy in the Doberman pinscher. Hum. Genet. 131, 1319–1325 (2012).
-
Meurs, Thou. M. et al. Clan of dilated cardiomyopathy with the striatin mutation genotype in boxer dogs. J. Vet. Intern. Med. 27, 1437–1440 (2013).
-
Meurs, K. M. et al. A missense variant in the titin cistron in Doberman pinscher dogs with familial dilated cardiomyopathy and sudden cardiac death. Hum. Genet. 138, 515–524 (2019).
-
Yost, O., Friedenberg, S. K., Jesty, Due south. A., Olby, North. J. & Meurs, K. M. The R9H phospholamban mutation is associated with highly penetrant dilated cardiomyopathy and sudden death in a spontaneous canine model. Gene 697, 118–122 (2019).
-
Meurs, Thousand. M. et al. Cess of PDK4 and TTN gene variants in 48 Doberman Pinschers with dilated cardiomyopathy. J. Am. Vet. Med. Assoc. 257, 1041–1044 (2020).
-
Van Vleet, J. F. & Ferrans, Five. J. Myocardial diseases of animals. Am. J. Pathol. 124, 98–178 (1986).
-
Packer, M. Cobalt cardiomyopathy: A critical appraisal in low-cal of a recent resurgence. Circ. Centre Fail. ix, e003604 (2016).
-
Weintraub, R. Grand., Semsarian, C. & Macdonald, P. Dilated cardiomyopathy. Lancet 390(10092), 400–414 (2017).
-
Reeves, W. C., Marcuard, Due south. P., Willis, S. Due east. & Movahed, A. Reversible cardiomyopathy due to selenium deficiency. J. Parent. Ent. Nutr. 13, 663–665 (1989).
-
Li, R. K., Sole, M. J., Mickle, D. A., Schimmer, J. & Goldstein, D. Vitamin East and oxidative stress in the center of the cardiomyopathic syrian hamster. Free Radic. Biol. Med. 24, 252–258 (1998).
-
Freeman, L. Thou. & Rush, J. E. Diet and cardiomyopathy: Lessons from spontaneous creature models. Curr. Heart Neglect. Rep. iv, 84–90 (2007).
-
Pion, P. D., Kittleson, Thousand. D., Rogers, Q. R. & Morris, J. G. Myocardial failure in cats associated with low plasma taurine: A reversible cardiomyopathy. Science 237, 764–768 (1987).
-
Pion, P. D., Kittleson, Chiliad. D., Thomas, West. P., Delellis, Fifty. A. & Rogers, Q. R. Response of cats with dilated cardiomyopathy to taurine supplementation. J. Am. Vet. Med. Assoc. 201, 275–284 (1992).
-
Kramer, G. A., Kittleson, Thousand. D., Fox, P. R., Lewis, J. & Pion, P. D. Plasma taurine concentrations in normal dogs and in dogs with heart disease. J. Vet. Intern. Med. ix, 253–258 (1995).
-
Kittleson, M. D., Keene, B., Pion, P. D. & Loyer, C. K. Results of the multicenter spaniel trial (MUST): Taurine- and carnitine-responsive dilated cardiomyopathy in American cocker spaniels with decreased plasma taurine concentration. J. Vet. Intern. Med. 11, 204–211 (1997).
-
Pion, P. D., Sanderson, S. 50. & Kittelson, M. D. The effectiveness of taurine and levocarnitine in dogs with heart affliction. Vet. Clin. North Am. Small Anim. Pract. 28, 1495–1514 (1998).
-
Freeman, L. M., Rush, J. E., Brownish, D. J. & Roudebush, P. Relationship betwixt circulating and dietary taurine concentrations in dogs with dilated cardiomyopathy. Vet. Ther. 2, 370–378 (2001).
-
Sanderson, Due south. L. et al. Furnishings of dietary fat and L-carnitine on plasma and whole blood taurine concentrations and cardiac office in salubrious dogs fed protein-restricted diets. Am. J. Vet. Res. 62, 1616–1623 (2001).
-
Fascetti, A. J., Reed, J. R., Rogers, Q. R. & Backus, R. C. Taurine deficiency in dogs with dilated cardiomyopathy: 12 cases (1997–2001). J. Am. Vet. Med. Assoc. 223, 1137–1141 (2003).
-
Backus, R. C. et al. Taurine deficiency in Newfoundlands fed commercially available consummate and balanced diets. J. Am. Vet. Med. Assoc. 223, 1130–1136 (2003).
-
Delaney, S. J., Kass, P. H., Rogers, Q. R. & Fascetti, A. J. Plasma and whole blood taurine in normal dogs of varying size fed commercially prepared nutrient. J. Anim. Physiol. Anim. Nutr. (Berl.) 87, 236–244 (2003).
-
Spitze, A. R., Wong, D. L., Rogers, Q. R. & Fascetti, A. J. Taurine concentrations in animal feed ingredients; cooking influences taurine content. J. Anim. Physiol. Anim. Nutr. (Berl.) 87, 251–262 (2003).
-
Torres, C. Fifty., Backus, R. C., Fascetti, A. J. & Rogers, Q. R. Taurine status in normal dogs fed a commercial diet associated with taurine deficiency and dilated cardiomyopathy. J. Anim. Physiol. Anim. Nutr. (Berl.) 87, 359–372 (2003).
-
Belanger, M. C., Ouellet, Thousand., Queney, Thousand. & Moreau, M. Taurine-scarce dilated cardiomyopathy in a family of gold retrievers. J. Am. Anim. Hosp. Assoc. 41, 284–291 (2005).
-
Backus, R. C. et al. Depression plasma taurine concentration in Newfoundland dogs is associated with low plasma methionine and cyst(e)ine concentrations and low taurine synthesis. J. Nutr. 136, 2525–2533 (2006).
-
Ko, K. S., Backus, R. C., Berg, J. R., Lame, Grand. W. & Rogers, Q. R. Differences in taurine synthesis rate among dogs relate to differences in their maintenance energy requirement. J. Nutr. 137, 1171–1175 (2007).
-
Greyness, Chiliad. et al. The result of 48-hour fasting on taurine condition in salubrious developed dogs. J. Anim. Physiol. Anim. Nutr. (Berl.) 100, 532–536 (2016).
-
Kaplan, J. 50. et al. Taurine deficiency and dilated cardiomyopathy in Golden Retrievers fed commercial diets. PLoS One. 13, e0209112 (2018).
-
Ontiveros, East. S. et al. Evolution of plasma and whole blood taurine reference ranges and identification of dietary features associated with taurine deficiency and dilated cardiomyopathy in golden retrievers: A prospective, observational study. PLoS Ane. 15, e0233206 (2020).
-
Keene, B. Westward. et al. Myocardial L-carnitine deficiency in a family of dogs with dilated cardiomyopathy. J. Am. Vet. Med. Assoc. 198, 647–650 (1991).
-
Rizos, I. 3-yr survival of patients with heart failure caused by dilated cardiomyopathy and L-carnitine assistants. Am. Center J. 139(2 Pt 3), S120–S123 (2000).
-
Kärkkäinen, O. et al. Whole grain intake associated molecule 5-aminovaleric acid, betaine decreases β-oxidation of fatty acids in mouse cardiomyocytes. Sci. Rep. 8, 13036 (2018).
-
Millard, H. R. et al. Dietary choline and betaine; associations with subclinical markers of cardiovascular disease risk and incidence of CVD, coronary middle affliction and stroke: The Jackson Heart Written report. Eur. J. Nutr. 57, 51–lx (2018).
-
Adin, D. et al. Echocardiographic phenotype of canine dilated cardiomyopathy differs based on diet blazon. J. Vet. Cardiol. 21, 1–9 (2019).
-
United states Food and Drug Administration. FDA investigating potential connections betwixt nutrition and cases of canine heart disease. https://www.fda.gov/animal-veterinary/cvm-updates/fda-investigating-potential-connection-between-nutrition-and-cases-canine-heart-disease (July 12, 2018).
-
United States Food and Drug Administration. FDA provides update on investigation into potential connectedness between sure diets and cases of canine eye disease. https://www.fda.gov/fauna-veterinarian/cvm-updates/fda-provides-update-investigation-potential-connection-between-certain-diets-and-cases-canine-heart. February 19, 2019.
-
United States Food and Drug Administration. FDA provides third status report on investigation into potential connection between sure diets and cases of canine center disease. https://www.fda.gov/fauna-veterinary/cvm-updates/fda-provides-tertiary-status-report-investigation-potential-connexion-between-certain-diets-and-cases. June 27, 2019.
-
Freid, K. J. et al. Retrospective study of dilated cardiomyopathy in dogs. J. Vet. Intern. Med. 35(1), 58–67 (2021).
-
Jones J., Carey Fifty., & Palmer L.A. FDA update on dilated cardiomyopathy: Fully and partially recovered cases. Scientific Forum Exploring Causes of Dilated Cardiomyopathy in Dogs. https://www.ksvdl.org/resources/documents/dcm-forum/FDA_KSU-Science-Forum-slides_09-29-2020.pdf. September 29, 2020.
-
Walker A. L. et. al. Association of diet with clinical outcomes in dogs with dilated cardiomyopathy and congestive center failure. J. Vet. Cardiol. S1760–2734(21)00019–nine (2021). Online ahead of print.
-
Solomon, South. K. Opening remarks. Scientific Forum Exploring Causes of Dilated Cardiomyopathy in Dogs. http://www.ksvdl.org/resource/documents/dcm-forum/DCM-Forum-SolomonOpening-Remarks.pdf. September 29, 2020.
-
Gibbons, H. & Brennan, L. Metabolomics equally a tool in the identification of dietary biomarkers. Proc. Nutr. Soc. 76, 42–53 (2017).
-
Zhang, 50. et al. Functional metabolomics characterizes a key role for North-acetylneuraminic acid in coronary avenue diseases. Circulation. 137, 1374–1390 (2018).
-
Kim, S., Kim, J., Yun, E. J. & Kim, One thousand. H. Food metabolomics: From farm to human. Curr. Opin. Biotechnol. 37, 16–23 (2016).
-
Montero, L. & Herrero, M. Ii-dimensional liquid chromatography approaches in foodomics - A review. Anal. Chim. Acta 1083, 1–18 (2019).
-
Dunn, Westward. A., Aronson, North. N. Jr. & Englard, S. The furnishings of ane-amino-D-proline on the product of carnitine from exogenous poly peptide-bound trimethyllysine past the perfused rat liver. J. Biol. Chem. 257, 7948–7951 (1982).
-
Roman-Garcia, P. et al. Vitamin B12-dependent taurine synthesis regulates growth and bone mass. J. Clin. Invest. 124, 2988–3002 (2014).
-
Markovich, J. E., Freeman, L. Thousand. & Heinze, C. R. Assay of thiamine concentrations in commercial canned foods formulated for cats. J. Am. Vet. Med. Assoc. 244, 175–179 (2014).
-
DiNicolantonio, J. J., Niazi, A. K., Lavie, C. J., O'Keefe, J. H. & Ventura, H. O. Thiamine supplementation for the treatment of heart failure: A review of the literature. Congest. Heart Fail. 19, 214–222 (2013).
-
Grundy, K. K. et al. Re-evaluation of the mechanisms of dietary fibre and implications for macronutrient bioaccessibility, digestion and postprandial metabolism. Brit. J. Nutr. 116, 816–833 (2016).
-
Wedekind, K. J. & Lowry, South. R. Are organic zinc sources efficacious in puppies?. J. Nutr. 128, 2593S-2595S (1998).
-
National Research Council. Minerals. In: Nutrient Requirements of Dogs and Cats. 145–192 (National Academies Printing, 2006).
-
Tamai, I. et al. Molecular and functional identification of sodium ion-dependent, high affinity human carnitine transporter OCTN2. J. Biol. Chem. 273, 20378–20382 (1998).
-
Grube, G. et al. Selective regulation of Cardiac Organic Cation Transporter Novel Type 2 (OCTN2) in dilated cardiomyopathy. Am. J. Pathol. 178, 2547–2559 (2011).
-
Wu, 10. et al. Functional characteristics and tissue distribution design of organic cation transporter 2 (OCTN2), an organic cation/carnitine transporter. J. Pharm. Exp. Ther. 290, 1482–1492 (1999).
-
Jones, D. A. Why are so many nutrient plants cyanogenic?. Phytochemistry 47, 155–162 (1998).
-
Alexander, D., Lombardi, R., Rodriguez, G., Mitchell, M. Grand. & Marian, A. J. Metabolomic stardom and insights into the pathogenesis of homo primary dilated cardiomyopathy. Eur. J. Clin. Invest. 41, 527–538 (2011).
-
Verdonschot, J. A. J. et al. Metabolic profiling assembly with disease severity in nonischemic dilated cardiomyopathy. J. Carte. Fail. 26, 212–222 (2020).
-
Posma, J. M. et al. Integrated analytical and statistical two-dimensional spectroscopy strategy for metabolite identification: Awarding to dietary biomarkers. Anal. Chem. 89, 3300–3309 (2017).
-
Madrid-Gambin, F. et al. Untargeted 1H NMR-based metabolomics analysis of urine and serum profiles later on consumption of lentils, chickpeas, and beans: An extended meal study to notice dietary biomarkers of pulses. J. Agric. Nutrient Chem. 66, 6997–7005 (2018).
-
Mohamadi, N., Sharififar, F., Pournamdari, M. & Ansari, Grand. A review on biosynthesis, analytical techniques, and pharmacological activities of trigonelline. J. Diet. Suppl. 15, 207–222 (2018).
-
Martin-Cabrejas, Chiliad. A. et al. Event of industrial aridity on the soluble carbohydrates and dietary fiber fractions in legumes. J. Agric. Nutrient Chem. 54, 7652–7657 (2006).
-
Njoumi, S., Josephe Amiot, Yard., Rochette, I., Bellagha, S. & Mousquet-Rivier, C. Soaking and cooking modify the blastoff-galacto-oligosaccharide and dietary fibre content in five Mediterranean legumes. Int. J. Nutrient Sci. Nutr. seventy, 551–561 (2019).
-
World Small Animate being Veterinary Association Global Nutrition Commission. Guidelines on selecting pet foods. https://wsava.org/wp-content/uploads/2021/04/Selecting-a-pet-food-for-your-pet-updated-2021_WSAVA-Global-Nutrition-Toolkit.pdf.
Acknowledgements
This work was funded in part past Nestlé Purina PetCare and the Barkley Fund. This work was funded in part by United States Section of Agriculture project number 8050-51000-107-00D, and this entity had no function in the design of the experiments, in the collection, analysis and interpretation of data, nor in composing the manuscript. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific data and does not imply recommendation or endorsement by the U.S. Department of Agronomics. The USDA is an equal opportunity provider and employer.
Writer information
Affiliations
Contributions
L.M.F. and J.Eastward.R. conceptualized the study. C.E.S., Fifty.D.P. and C-Q.Fifty contributed to data analysis. C.Eastward.S., L.D.P., C-Q.L., J.E.R. and L.K.F. wrote and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
In the last three years, Dr. Freeman has received research funding from, given sponsored lectures for, and/or provided professional services to Aratana Therapeutics, Elanco, Hill's Pet Nutrition, Nestlé Purina PetCare, P&M Pet Care (now Mars), and Royal Canin. In the last 3 years, Dr. Rush has received research funding from, given sponsored lectures for, and/or provided professional services to Aratana Therapeutics, Boehringer Ingelheim, Elanco, IDEXX, Nestlé Purina PetCare, and Royal Canin. None of the other authors has any competing interests to declare.
Additional information
Publisher's notation
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Eatables Attribution 4.0 International License, which permits employ, sharing, accommodation, distribution and reproduction in any medium or format, as long equally you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and betoken if changes were made. The images or other third party fabric in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the textile. If material is not included in the article's Artistic Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted employ, yous volition demand to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
About this article
Cite this article
Smith, C.E., Parnell, Fifty.D., Lai, CQ. et al. Investigation of diets associated with dilated cardiomyopathy in dogs using foodomics analysis. Sci Rep 11, 15881 (2021). https://doi.org/x.1038/s41598-021-94464-two
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/s41598-021-94464-ii
Comments
By submitting a annotate you agree to abide by our Terms and Customs Guidelines. If y'all find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Source: https://www.nature.com/articles/s41598-021-94464-2
Post a Comment for "Taurine Deficiency Dilated Cardiomyopathy in a Family of Golden Retrievers"